题目
多项填空题
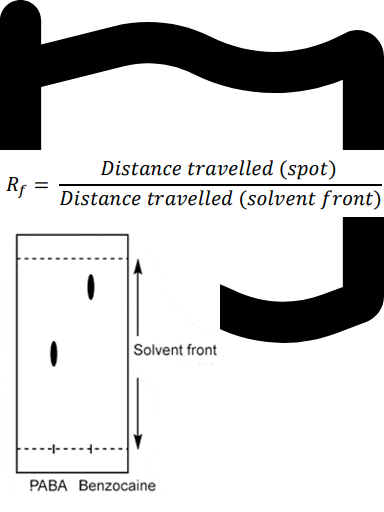
Question textAn example of Thin Layer Chromatography (TLC) results: Solvent used in developing a TLC plate: 50:50 hexane : ethyl acetate. Calculate the Retention factor (Rf) value for each component (ρ-aminobenzoic acid (PABA) and Benzocaine) on the plate. (to 2 significant figures) Please refer to p.47 in the lab manual for more information. [table] TLC results | Rf (retention factor) ρ-aminobenzoic acid (PABA):Distance travelled: 2.1 cm Benzocaine: Distance travelled: 3.5 cm Solvent front: Distance travelled: 4.1 cm | Rf (PABA)= Answer 1 Question 6 Rf (Benzocaine)= Answer 2 Question 6 [/table] Based on the information above, which compound is less polar? Multiple choice 1 Question 61. PABA2. Benzocaine3. NeitherMark 1.00 out of 1.00
查看解析
标准答案
Please login to view
思路分析
Let’s break down the data given for the TLC results and then interpret what it means for polarity.
First, we calculate the Rf values using the formula: Rf = distance travelled by compound / distance travelled by solvent front.
For ρ-aminobenzoic acid (PABA): distance travelled = 2.1 cm, sol......Login to view full explanation登录即可查看完整答案
我们收录了全球超50000道考试原题与详细解析,现在登录,立即获得答案。
类似问题
Question textAn example of Thin Layer Chromatography (TLC) results:Solvent used in developing a TLC plate: 50:50 hexane : ethyl acetate.Calculate the Retention factor (Rf) value for each component (ρ-aminobenzoic acid (PABA) and Benzocaine) on the plate. (to 2 significant figures)Please refer to p.46 in the lab manual for more information.[table] TLC results | Rf (retention factor) ρ-aminobenzoic acid (PABA):Distance travelled: 2.1 cmBenzocaine: Distance travelled: 3.5 cmSolvent front: Distance travelled: 4.1 cm | Rf (PABA)= Answer 1 Question 6 Rf (Benzocaine)= Answer 2 Question 6 [/table]Based on the information above, which compound is less polar? Answer 3 Question 6[select: , Benzocaine, PABA, Neither]
Q6 V3Consider the following TLC plate of compounds X, Y and Z developed using a suitable mobile phase on a polar stationary phase. Which of the following is correct?
Question textAn example of Thin Layer Chromatography (TLC) results: Solvent used in developing a TLC plate: 50:50 hexane : ethyl acetate. Calculate the Retention factor (Rf) value for each component (ρ-aminobenzoic acid (PABA) and Benzocaine) on the plate. (to 2 significant figures) Please refer to p.47 in the lab manual for more information. [table] TLC results | Rf (retention factor) ρ-aminobenzoic acid (PABA):Distance travelled: 2.1 cm Benzocaine: Distance travelled: 3.5 cm Solvent front: Distance travelled: 4.1 cm | Rf (PABA)= Answer 1 Question 6 Rf (Benzocaine)= Answer 2 Question 6 [/table] Based on the information above, which compound is less polar? Multiple choice 1 Question 61. PABA2. Benzocaine3. NeitherMark 1.00 out of 1.00
Samples of three amino acids A, B and C, were spotted on the base line of a TLC plate. The chromatogram produced using a particular solvent is shown below: Amino acids Rf values alanine 0.56 isoleucine 0.79 taurine 0.34 Use the TLC plate and Rf values given to identify amino acid A.
更多留学生实用工具
希望你的学习变得更简单
加入我们,立即解锁 海量真题 与 独家解析,让复习快人一步!